
30 Years of Revolutionizing Home Cooking
We explore the science behind what makes a recipe tick. We ask questions, conduct experiments, and get answers—delicious answers that you won't find anywhere else.

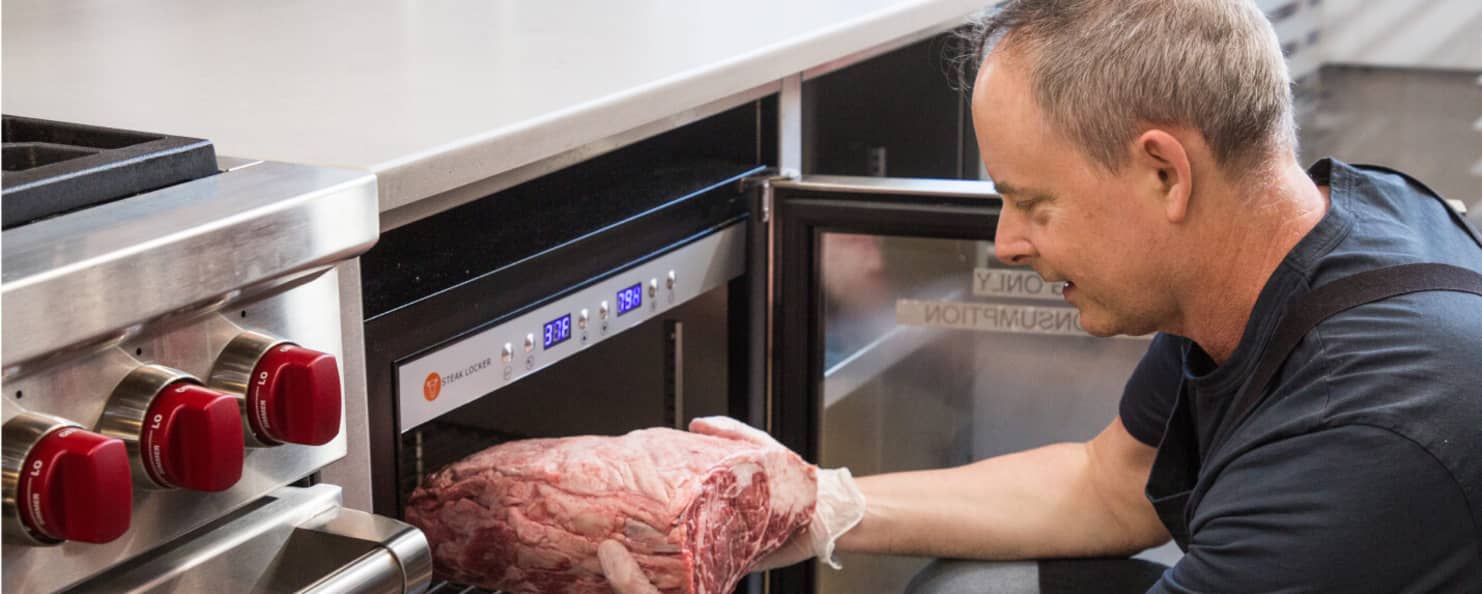


Join Us in the Lab
Good science makes great food
We believe that a little science goes a long way to helping you understand what's going on in your food and in your kitchen. You'll learn why a technique works or how an ingredient creates an important effect.
Can Baking Soda Remove Pesticides from Produce?
A study found that soaking produce in baking soda solution can remove common pesticides. We conducted our own experiment to see how it compares to other common methods.
Can You Cook a Frozen Steak?
Conventional wisdom holds that frozen steaks should be thawed before cooking. Our testing proved otherwise. Learn why forgetting to defrost steak ahead of time isn’t necessarily a bad thing.
Why Very Hot and Very Cold Foods Need More Salt
Serving temperature of food dramatically affects how seasoned it tastes. The more extreme the temperature—whether piping hot or very chilled—the more salt you need. We ran an experiment to prove it.
Temper Chocolate with a Microwave and a Box Grater
Forget the fussy heat-cool-reheat method. With just a microwave and a box grater you can create shiny, snappy chocolate for drizzling, dipping, or coating in no time.
Ultimate Charcoal-Grilled Steaks
The secret to delivering a charcoal-grilled steak with a killer crust and perfectly cooked meat from edge to edge? Keep the coals in the chimney and think outside the grill.
Caramelized Onions
The sweetest, most complex caramelized onions usually take an hour of careful cooking over a low flame. We cut that time in half with an ingredient we least expected: water.
Perfect Scrambled Eggs
The classic low-heat approach will never give you tender scrambled eggs with big, pillowy curds. But achieving scrambled egg perfection takes more than just turning up the heat.
The Best Baked Potato
We wanted a uniformly fluffy interior encased in thin, crisp skin—and we baked nearly 200 pounds of potatoes to get it. The key to our success? An instant-read thermometer.Our Signature Process
We put every recipe through the same rigorous process, testing and retesting every variable and then taking what we've learned to create a dish that's the very best in its class.
1. The Five-Recipe Test
First we seek out five existing recipes. We taste and assess them, noting qualities we like and any we don’t.

2. Our First Draft
Next, we define goals for our version, and sketch out the most promising way to reach them.

3. Test, Taste, Repeat
Over at least six weeks, we test and retest every recipe variable, learning from our mistakes so we can pass on that information to you.

4. Final, Failproof Results
At last—a recipe that works perfectly in our kitchen and has been tested and approved by thousands of home testers.

Our Innovative Content Is Everywhere You Are

Access 30 Years of Our Award-Winning Magazine
Our foolproof recipes, explorations into food science, and thoroughly researched stories set new standards for what a cooking magazine can be.
Weeknight Roast Chicken

Perfect Chocolate Chip Cookies

Grilled Boneless Beef Short Ribs

Skillet-Roasted Brussels Sprouts with Chile, Peanuts, and Mint

New York–Style Thin-Crust Pizza

Dakganjeong (Korean Fried Chicken Wings)

Braised Asparagus with Lemon and Chives

Easy-Peel Hard-Boiled Eggs

Spaghetti All'Assassina

Murgh Makhani (Indian Butter Chicken)

Salt-Cured Egg Yolks

Easiest-Ever Biscuits
What's Eating Dan?
Cook’s Illustrated Editor in Chief Dan Souza goes deep on his favorite ingredients and dishes in this smart, fun series dedicated to understanding the science of good cooking.Techniquely with Lan Lam
Veteran restaurant cook and chemistry major, Cook’s Illustrated Senior Editor Lan Lam delves into some of our most surprising—and even counterintuitive—techniques, including cold-searing steak and overcooking vegetables.
Why You Should Trust Our Recipes
We’re the flagship brand behind America’s Test Kitchen's rigorous and scientific recipe development process. We make recipes that work—no hit or miss, no drama. Our recipes have been prodded, tweaked, and perfected until they're failproof in any kitchen.



“Recipes that always, always work”
- The New York Times



























